 1 Silver exists as silver metal and silver ions of different oxidation states of +1, +2, +3, and +4 that the most common states of silver are silver(0) metal and silver(-) ion and both of them interact with thiols in no redox reaction involved that Ag 2+, Ag 3+,and Ag 4+ form state are not of relevance for aqueous solutions and under environmental and biological conditions. 1 Released biologically active Ag + shows a strong affinity for sulphydryl groups and other anionic ligands of proteins, cell membranes, and debris that Ag + binds protein residues on cell membranes of sensitive bacteria and is absorbed intracellularly by pinocytosis which concentration of 60 ppm Ag + should be sufficient to control the majority of bacterial pathogen. Increasing use of silver as an efficacious chemotherapeutic antibacterial and antifungal agent in wound care products, medical devices textiles, cosmetics, and even domestic appliances in recent years has led to concern as to the safety aspects of the metal and potential risks associated with the absorption of the biologically acting Ag + into the human body. Silver of transition metal has highly antibacterial activities and is widely utilized as chemotherapy agents. Keywords: Silver (-) ions, PGN cell wall, outer membrane lipoproteins, bacteriolysis, hydrolase and degradation, PGN synthesis and autolysin, reactive oxygen species (ROS), DNA base-pairs Abbreviations DNA damages may be due to Ag +-coordinated complex formations by Ag + substitution within double and triple hydrogen bonds in DNA base pairs. Silver ions induced ROS generations such as O 2 -, H 2O 2,・OH, OH -producing in bacterial cell wall occur and lead to oxidative stress. coli cell wall by Ag + ions are caused by the destruction of outer membrane structure due to degradative enzymes of lipoproteins at N- and C-terminals, and by the inhibition of PGN elongation owing to inactivation of PGN TP synthetic enzyme endopeptidase and enhancement of the activations of PGN hydrolases and autolysins of Amidase, Peptidase, and Carboxypeptidase. On the other hand, bacteriolysis and destruction against E. Bacteriolysis against S.aureus peptidoglycan (PGN) cell wall by Ag + ions is due to inhibition of PGN elongation caused by regulation of PGN synthetic transglycosylase (TG) and transpeptidase (TP), and enhancement of the activation of PGN autolysins of Amidases. In some systems with high ion activity (e.g., saline soils, fertilizer bands), net adsorption may be limited by the capacity of the PRS® Probe to hold ions.Antibacterial mechanism of bacteriolyses and destructions of bacterial cell walls by silver(Ⅰ) ions has been considered against Staphylococcus aureus (S. Conversely, declining activities of a strongly held ion may not result in the amount adsorbed by PRS® Probes unless the activity and affinity of other ions is sufficient to displace the strongly held ion. Increasing activity of an ion that is strongly held will generally increase the quantity of that ion on PRS® Probes and reduce the quantity of ions that are weakly held. 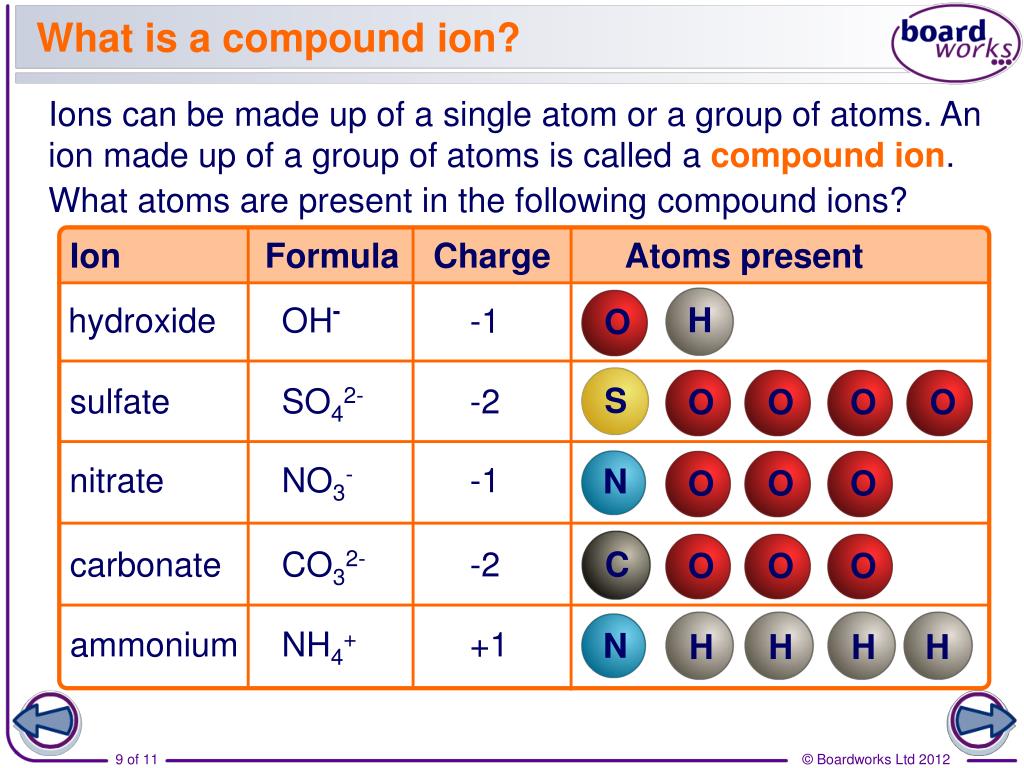
The principles of ion exchange continue to apply. Soils are constantly undergoing change, and therefore the quantity of ions adsorbed on PRS® Probes may also change. Within a given valence, ions that have a smaller dehydrated radius are generally held more strongly. Relative affinity of an ion on a resin membrane (or other soil colloids) is most strongly influenced by valence: divalent ions are retained more strongly than monovalent ions, and trivalent ions are retained even more strongly. In both systems, a critical rate-limiting step is diffusion to the sink in response to gradients established by dynamic equilibria.Įventually, the quantity of a specific ion adsorbed on the ion exchange membrane often reaches a constant level that is Equilibrium Dependent, it depends on the relative activity in soil solution and relative affinity on the membrane of the ion. 
The process is similar to that occurring in the soil-root system, in which roots adsorb nutrients from soil solution by releasing counter ions such as H +, OH -, and HCO 3. When PRS® Probes are first inserted in soil, the rate of exchange is Diffusion Dependent: the flux of ions to the ion exchange membrane is controlled by the activity and diffusivity of ions in soil solution. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
